Abstract
Introduction: Mutations involving isocitrate dehydrogenase 1/2 (IDH) are known oncogenic drivers in hematological malignancies, conferring neomorphic enzymatic activity to IDH 1/2, resulting in the oncometabolite, 2-hydroxyglutarae (2-HG). 2-HG in turn suppresses TET activity, making IDH and TET2 mutations synthetically lethal and almost mutually exclusive. The frequency of IDH mutations in CMML is <10% and their prognostic impact remains unclear. We carried out this study in a large database of molecularly annotated CMML patients to better define the clinical profile and prognostic impact of these mutations.
Methods: After IRB approval, CMML patients from the Mayo Clinic, Minnesota and the Moffitt Cancer Center (MCC), Tampa, Florida, were included in the study. All patients had bone marrow (BM) biopsies with cytogenetics and molecular genetics done either at diagnosis, or at first referral. Clinical and mutational data were abstracted and retrospectively analyzed. Overall survival (OS) was calculated from date of CMML diagnosis to date of death/last follow, while AML-free survival (AML-FS) was calculated from date of CMML diagnosis to date of leukemic transformation (LT). Patients that had undergone allogeneic HCT were excluded from the study (n=3). Statistical analysis was carried out using the Blue Sky software.
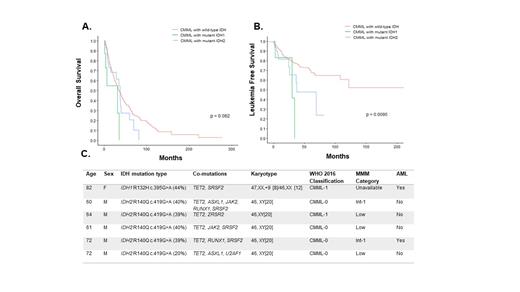
Results: Six hundred and forty four patients were included in the study (Mayo Clinic-357, MCC- 287), median age 71 years (range, 20-95 years), 67.8% being male. Forty-three (6.7%) patients had IDH mutations, 35 (82%) IDH2 and 8 (18%) IDH1; of which, 34 (97%) involved the IDH2R140 hotspot and 5 (62.5%) involved the IDH1R132 hotspot, respectively. The median variant allele fractions (VAF) for IDH1 mutations was 41% (range, 8-46%) and for IDH2 mutations was 46% (range, 7-70%). There were no significant demographic or clinical differences between IDH mutant and wild type CMML patients, with the exception that IDH mutant CMML patients were less likely to be thrombocytopenic (p=0.006), were less likely to have TET2 co-mutations (14% vs 53.2%; p<0.001) and were more likely to have SRSF2 co-mutations (69.8% VS 40.3%; p<0.001). Importantly there were no differences in proliferative or dysplastic subtypes (p=0.3), cytogenetic (p=0.12) and molecular risk stratifications (p=045). There were also no significant demographic or clinical differences between IDH1 vs IDH2 mutant CMML patients. Six (14%) IDH mutant CMML patients had TET2 co-mutations; 5 (83%) with IDH2R140Q (median VAF-28%;all male) and 1 (17%) with IDH1R132H (VAF-44%;female) (Figure 1). Five (11%) IDH2 mutant patients were treated with enasidenib (IDH2 inhibitor), none with a durable response, while none of the IDH1 mutant patients received targeted therapy.
At last follow up (median 18 months), 337 (52%) deaths and 119 (18.5%) LT have been documented, with IDH mutant patients having a higher LT rate (30.2% vs 17.6%, p=0.04) compared to wildtype patients. The median OS of the entire cohort was 35 months, with no difference in OS between IDH mutant and wild type patients (34.5 vs 35 months, p=0.12), with IDH1 mutant patients having a shorter OS in comparison to IDH2 mutant patients (31 vs 37 months; p=0.005- Figure 1). IDH mutant CMML patients also had a shorter AML-FS in comparison to wild type patients (36.6 vs 210 months, p=0.005), with there being no differential impact on AML-FS of IDH1 vs IDH2 mutations (p=0.26, Figure 1).
Conclusions: IDH mutations are infrequent in CMML (7%), with IDH2 mutations being more common than IDH1 mutations (80 vs 20%). IDH mutations co-occur very infrequently with TET2 mutations (14%), with IDH mutant patients being less likely to have thrombocytopenia and more likely to have SRSF2 co-mutations. IDH mutations negatively impacting AML-FS without a significant impact on OS. Prospective clinical trials testing the safety and efficacy of IDH1/2 inhibitors in CMML are much needed.
Komrokji: AbbVie: Consultancy; PharmaEssentia: Membership on an entity's Board of Directors or advisory committees; Taiho Oncology: Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Jazz: Consultancy, Speakers Bureau; Acceleron: Consultancy; BMSCelgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Geron: Consultancy. Al-Kali: Novartis: Research Funding; Astex: Other: Research support to institution. Padron: BMS: Research Funding; Stemline: Honoraria; Taiho: Honoraria; Kura: Research Funding; Incyte: Research Funding; Blueprint: Honoraria. Patnaik: StemLine: Research Funding; Kura Oncology: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal